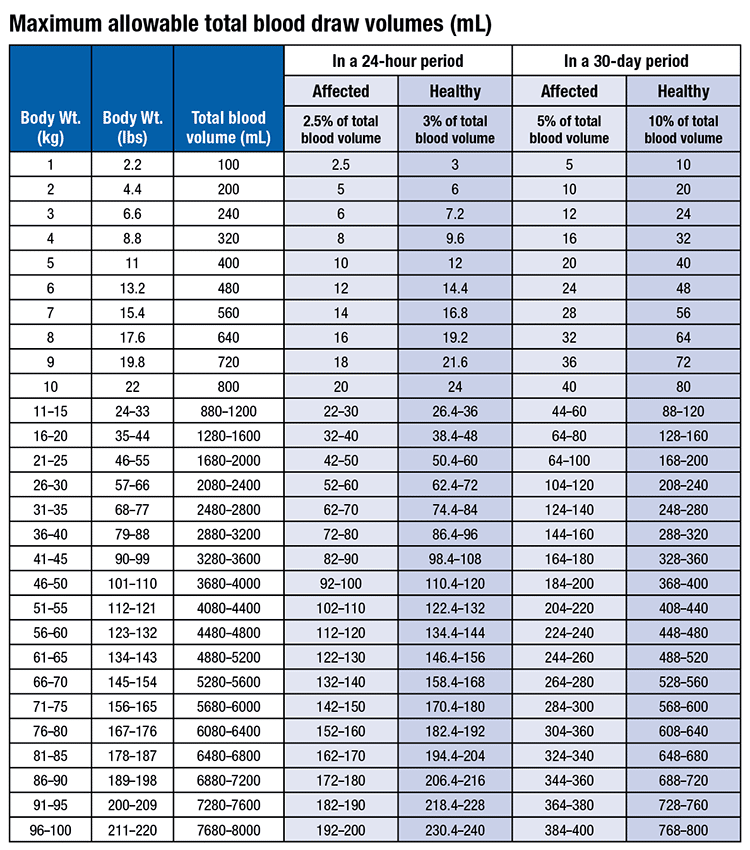
ACCEPTABLE BLOOD VOLUMES DRAWN FOR CHILREN IN RESEARCH STUDIES CHLA IRB Blood Draw Volume Policy CHLA IRB policy is to limit th

Table 2 from Blood sample volumes in child health research: review of safe limits. | Semantic Scholar

Blood draws up to 3% of blood volume in clinical trials are safe in children - Peplow - 2019 - Acta Paediatrica - Wiley Online Library



:max_bytes(150000):strip_icc()/tips-for-making-a-blood-draw-easier-3156931-a1efe64eb50b4ca3b489144274854bf0.png)