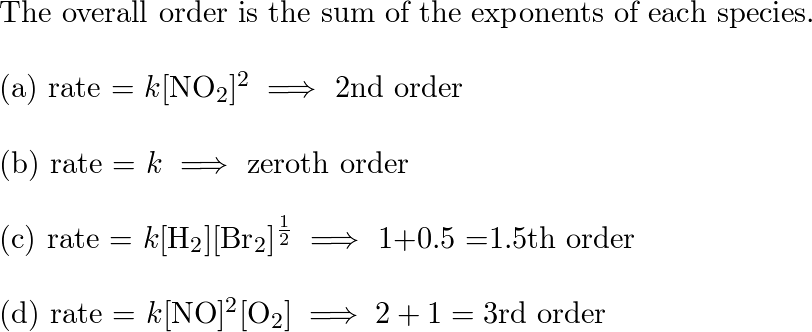SOLVED: The reaction below has a rate constant of 2.05××10−−4 s-1 at a specific temperature. C2H4O(g) → CH4(g) + CO(g) What is the overall order of the reaction? a Zero Order b
![SOLVED: Give the individual reaction orders for all substances and the overall reaction order from this rate law: rate = k [ H N O 2 ] 4 [ N O ] SOLVED: Give the individual reaction orders for all substances and the overall reaction order from this rate law: rate = k [ H N O 2 ] 4 [ N O ]](https://cdn.numerade.com/ask_previews/2bea17b9-3891-44d9-82f8-c1ad67dda6c7_large.jpg)
SOLVED: Give the individual reaction orders for all substances and the overall reaction order from this rate law: rate = k [ H N O 2 ] 4 [ N O ]
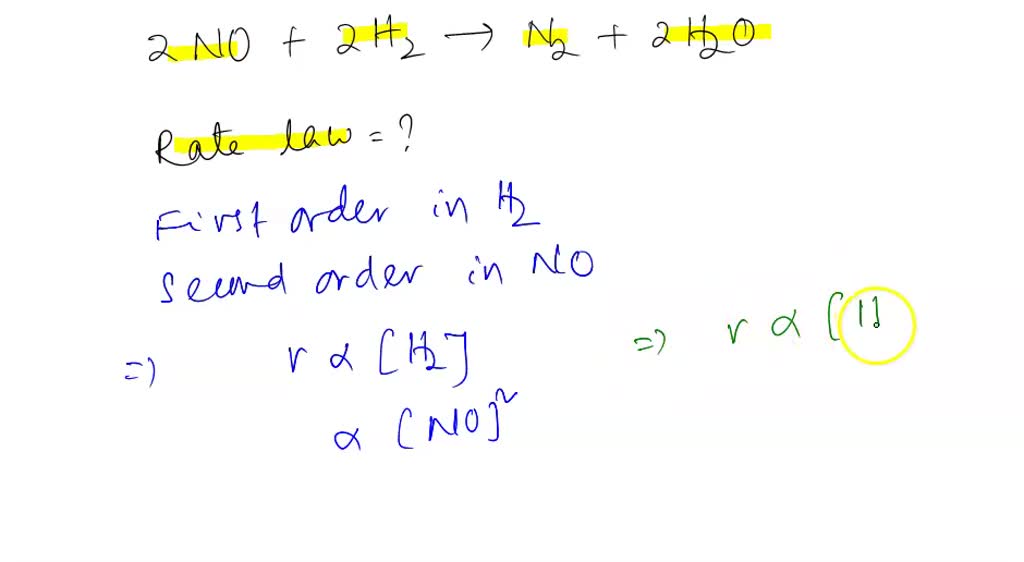
SOLVED: 1a. The gas phase reaction of hydrogen with iodine H2 + I2 2 HI is first order in H2 and second order overall. Complete the rate law for this reaction in

Amazon.com : Herm Sprenger Chrome-Plated Steel Choke Dog Training Collar with Toggle-Closure No-Pull Slip Chain for Small Medium Large Dogs for Easy Control Show Chain Collar Made in Germany : Pet Supplies

Steelers Depot 7⃣ on Twitter: "2023 NFL Draft order: Bears, Texans still in play for No. 1 overall pick https://t.co/N3XnL8vDiC #Steelers https://t.co/kP3CYU32j7" / Twitter
![SOLVED: What is the overall order of the reaction below CO (g) + NO2 (g) -> CO2 (g) + NO (g) if it proceeds via the following rate expression? Rate = k[CO][NO2] SOLVED: What is the overall order of the reaction below CO (g) + NO2 (g) -> CO2 (g) + NO (g) if it proceeds via the following rate expression? Rate = k[CO][NO2]](https://cdn.numerade.com/ask_previews/c6b4e51-6361-5d18-dd24-a5f8efe7d_large.jpg)
SOLVED: What is the overall order of the reaction below CO (g) + NO2 (g) -> CO2 (g) + NO (g) if it proceeds via the following rate expression? Rate = k[CO][NO2]
![SOLVED: Classify each rate law based on whether the reaction is first-order; second-order; O third-order overall: First-order Second-order Third-order Answer Bank rate k[HCN] rate k[O] [NO] [NzE rate [BFs] [NH,] rate kINO]? SOLVED: Classify each rate law based on whether the reaction is first-order; second-order; O third-order overall: First-order Second-order Third-order Answer Bank rate k[HCN] rate k[O] [NO] [NzE rate [BFs] [NH,] rate kINO]?](https://cdn.numerade.com/ask_previews/7a671ad0-bb32-4650-a5e2-0dc410a20fb1_large.jpg)
SOLVED: Classify each rate law based on whether the reaction is first-order; second-order; O third-order overall: First-order Second-order Third-order Answer Bank rate k[HCN] rate k[O] [NO] [NzE rate [BFs] [NH,] rate kINO]?

X-Large Black Polyester / Polyurethane Single Needle Stitched No Pockets Adjustable Suspenders / Snaps High Visibility Overall | Fastenal